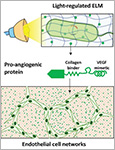
Regenerative medicine aims to restore damaged cells, tissues, and organs, for which growth factors are vital to stimulate regenerative cellular transformations. Major advances have been made in growth factor engineering and delivery like the development of robust peptidomimetics and controlled release matrices. However, their clinical applicability remains limited due to their poor stability in the body and need for careful regulation of their local concentration to avoid unwanted side-effects. In this study, a strategy to overcome these limitations is explored using engineered living materials (ELMs), which contain live microorganisms that can be programmed with stimuli-responsive functionalities. Specifically, the development of an ELM that releases a pro-angiogenic protein in a light-regulated manner is described. This is achieved by optogenetically engineering bacteria to synthesize and secrete a vascular endothelial growth factor peptidomimetic (QK) linked to a collagen-binding domain. The bacteria are securely encapsulated in bilayer hydrogel constructs that support bacterial functionality but prevent their escape from the ELM. In situ control over the release profiles of the pro-angiogenic protein using light is demonstrated. Finally, it is shown that the released protein is able to bind collagen and promote angiogenic network formation among vascular endothelial cells, indicating the regenerative potential of these ELMs.